Acid rain describes the acidification of rain by airborne acids. These acids are commonly produced by mad made sources, particularly combustion of fossil fuels in power plants and cars and trucks, but it can also be produced by sources in nature, such as volcanoes. Acid rain is a serious environmental problem because it is harmful to plant life and water life, such as in lakes and streams. t Acid rain is particularly damaging to lakes, streams, and forests and the plants and animals that live in these ecosystems in large parts of the United States ,Canada and China. First, here is information for the general public, then below is information that environmental professionals may need:
You can also find information about acid rain where you live , numerous educational resources , and other sources of information on acid rain.
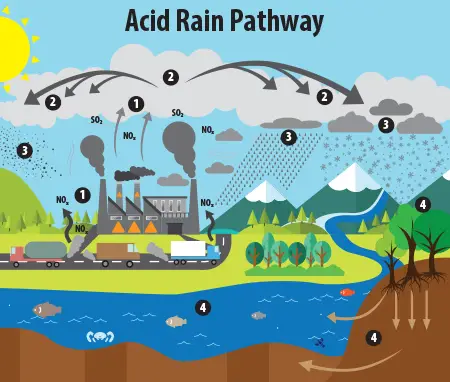
Acid rain results when sulfur dioxide (SO2) and nitrogen oxides (NOX) are emitted into the atmosphere and transported by wind and air currents. The SO2 and NOX react with water, oxygen and other chemicals to form sulfuric and nitric acids. These then mix with water and other materials before falling to the ground.
While a small portion of the SO2 and NOX that cause acid rain is from natural sources such as volcanoes, most of it comes from the burning of fossil fuels. The major sources of SO2 and NOX in the atmosphere are:
Winds can blow SO2 and NOX over long distances and across borders making acid rain a problem for everyone and not just those who live close to these sources.
Wet deposition is what we most commonly think of as acid rain. The sulfuric and nitric acids formed in the atmosphere fall to the ground mixed with rain, snow, fog, or hail.
Acidic particles and gases can also deposit from the atmosphere in the absence of moisture as dry deposition. The acidic particles and gases may deposit to surfaces (water bodies, vegetation, buildings) quickly or may react during atmospheric transport to form larger particles that can be harmful to human health. When the accumulated acids are washed off a surface by the next rain, this acidic water flows over and through the ground, and can harm plants and wildlife, such as insects and fish.
The amount of acidity in the atmosphere that deposits to earth through dry deposition depends on the amount of rainfall an area receives. For example, in desert areas the ratio of dry to wet deposition is higher than an area that receives several inches of rain each year.
 Acidity
and alkalinity are measured using a pH scale for which 7.0 is neutral. The lower a substance's pH (less than 7), the more acidic it is; the higher a
substance's pH (greater than 7), the more alkaline it is. Normal rain has a pH of about 5.6; it is slightly acidic because carbon dioxide (CO2)
dissolves into it forming weak carbonic acid. Acid rain usually has a pH between 4.2 and 4.4.
Acidity
and alkalinity are measured using a pH scale for which 7.0 is neutral. The lower a substance's pH (less than 7), the more acidic it is; the higher a
substance's pH (greater than 7), the more alkaline it is. Normal rain has a pH of about 5.6; it is slightly acidic because carbon dioxide (CO2)
dissolves into it forming weak carbonic acid. Acid rain usually has a pH between 4.2 and 4.4.
Policymakers, research scientists, ecologists, and modelers rely on the National Atmospheric Deposition Program’s (NADP) National Trends Network (NTN) for measurements of wet deposition. The NADP/NTN collects acid rain at more than 250 monitoring sites throughout the US, Canada, Alaska, Hawaii and the US Virgin Islands. Unlike wet deposition, dry deposition is difficult and expensive to measure. Dry deposition estimates for nitrogen and sulfur pollutants are provided by the Clean Air Status and Trends Network (CASTNET). Air concentrations are measured by CASTNET at more than 90 locations.
When acid deposition is washed into lakes and streams, it can cause some to turn acidic. The Long-Term Monitoring (LTM) Network measures and monitors surface water chemistry at over 280 sites to provide valuable information on aquatic ecosystem health and how water bodies respond to changes in acid-causing emissions and acid deposition.
Next, learn about the Effects of Acid Rain.
Or, learn more about:
Ways to save money AND help the environment:
Save water AND money with this showerhead adapter, it lets the water flow until the water is hot, then shuts off water flow until you restart it, ShowerStart TSV Hot Water Standby Adapter
it lets the water flow until the water is hot, then shuts off water flow until you restart it, ShowerStart TSV Hot Water Standby Adapter

Protect your health with these:
Mattress Dust mite-Bedbug protector, 100% Waterproof, Hypoallergenic, Zippered
Handheld Allergen Vacuum Cleaner with UV Sanitizing for Allergies and Pet, Kills Mite, Virus, Molds, True HEPA with Powerful Suction removes Hair, Dander, Pollen, Dust ,
,
Immune Support Supplement with Quercetin, Vitamin C, Zinc, Vitamin D3
GermGuardian Air Purifier with UV-C Light and HEPA 13 Filter, R emoves 99.97% of Pollutants
emoves 99.97% of Pollutants
HEPA Air Purifier, HEPA Air Cleaner with Washable Pre-Filter, AQI Display, ECO Mode, Sleep Mode, Pet Mode for Pets, Dust, Pollen, Removes 99.97% of Particles, Smoke, Mold, Pet Dander, Dust, Odors
Interesting Reads:
THE PREPPER'S CANNING & PRESERVING BIBLE: [13 in 1] Your Path to Food Self-Sufficiency. Canning, Dehydrating, Fermenting, Pickling & More, Plus The Food Preservation Calendar for a Sustainable Pantry
The Backyard Homestead: Produce all the food you need on just a quarter acre! P aperback
aperback
The Citizens' Guide to Geologic Hazards: A Guide to Understanding Geologic Hazards Including Asbestos, Radon, Swelling Soils, Earthquakes, Volcanoes
The Uninhabitable Earth: Life After Warming
Book: The Sixth Extinction:
An Unnatural History Paperback