According to NOAA, scientists estimate that 50 to 80% of the oxygen produced on Earth comes from the ocean. The majority of this production is from oceanic plankton; drifting plants, algae, and some bacteria that can photosynthesize; absorbing atmospheric carbon dioxide which dissolves in the oceans. One particular species, Prochlorococcus, is the smallest photosynthetic organism on Earth. But this little bacteria produces up to 20% of the oxygen in our entire biosphere. And as important tropical rainforests on land are to biodiversity, these little organisms produce more oxygen than all of the rain forests combined.
Calculating the exact percentage of oxygen produced in the ocean is difficult because the amounts are constantly changing. Scientists can use satellite imagery to track photosynthesizing plankton and estimate the amount of photosynthesis occurring in the ocean, but satellite imagery cannot tell the whole story. The amount of plankton changes seasonally and in response to changes in the water’s nutrient load, temperature, and other factors. Studies have shown that the amount of oxygen in specific locations varies with time of day and with the tides.
Ocean produces at least 50% of the oxygen on Earth, but some is consumed by marine life. Like animals on land, marine animals use oxygen to breathe, and both plants and animals use oxygen for cellular respiration. Oxygen is also consumed when dead plants and animals decay in the ocean.
This is particularly problematic when algal blooms die and the decomposition process uses oxygen faster than it can be replenished. This can create areas of extremely low oxygen concentrations, or hypoxia. These areas are often called dead zones, because the oxygen levels are too low to support most marine life. NOAA’s National Centers for Coastal Ocean Science conducts extensive research and forecasting on algal blooms and hypoxia to lessen the harm done to the ocean ecosystem and human environment..
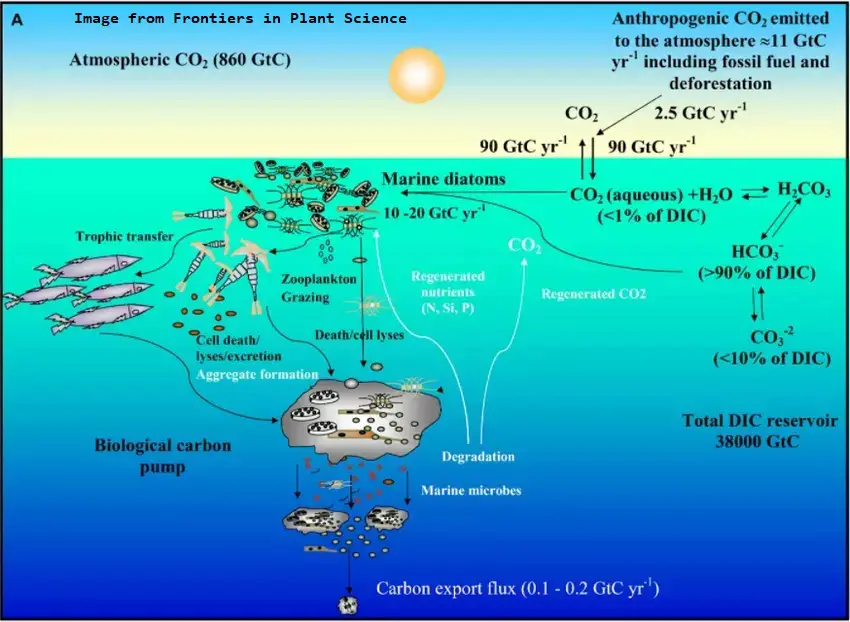
Just as the earth's oceans are vital to producing the oxygen we need, they are equally important in removing carbon in the form of carbon dioxide from the atmosphere. About 50 Gt (50 billion metric tons) of carbon is drawn down into the biological pump per year.
The University of Waikato Te Whare Wānanga o Waikato tells us:
Carbon dioxide from the atmosphere dissolves in the surface waters of the ocean. Some of the carbon dioxide stays as dissolved gas, but much of it gets turned into other things. Photosynthesis by tiny marine plants (phytoplankton) in the sunlit surface waters turns the carbon into organic matter. Many organisms use carbon to make calcium carbonate, a building material of shells and skeletons. Other chemical processes create calcium carbonate in the water. The using up of carbon by biological and chemical processes allows more carbon dioxide to enter the water from the atmosphere.
The shells and skeletons of these tiny organisms like brachiopods, mollusks, diatoms, echinoids, coccolithophores, foraminifera and corals builds up on the ocean floor. Some dissolve, but most build up slowly on the sea floor forming calcium carbonate (CaCO3) sediments. The layers compact as the shells of calcite fragments press down on top of each other. Eventually, tectonic processes of high heat and pressure transform these sediments into limestone. This process locks huge amounts of carbon away from the atmosphere for millions of years.
The White Cliffs of Dover on the coast of England are an example of limestone from calcium carbonate sediments that were deep under the ocean millions of years ago.
The World Economic Forum says:
Diatoms are not the only marine plant life capable of absorbing carbon. Seagrass - which covers just 0.2% of the seabed, but accounts for 10% of the ocean’s capacity to store carbon - could play a key role in slowing climate change.
A small percentage of the earth's limestone is formed through a non-biologic of evaporation , as the water containing the dissolved calcium carbonate precipitates and is deposited
Limestone is currently forming in the Caribbean Sea, Bahamas, Indian Ocean, Persian Gulf, Gulf of Mexico, around Pacific Ocean islands and the Indonesian archipelago.
To give you an idea of the vast magnitude of the carbon dioxide that is removed by these little creatures, almost all of the limestone formations on earth are made up of the shells of these tiny animals.
Ocean acidification is a problem that could affect the carbon and oxygen cycles, as oceans acidify and corals and other ocean life are killed by it.
Ways to save money AND help the environment:
Save water AND money with this showerhead adapter, it lets the water flow until the water is hot, then shuts off water flow until you restart it, ShowerStart TSV Hot Water Standby Adapter
it lets the water flow until the water is hot, then shuts off water flow until you restart it, ShowerStart TSV Hot Water Standby Adapter

Protect your health with these:
Mattress Dust mite-Bedbug protector, 100% Waterproof, Hypoallergenic, Zippered
Handheld Allergen Vacuum Cleaner with UV Sanitizing for Allergies and Pet, Kills Mite, Virus, Molds, True HEPA with Powerful Suction removes Hair, Dander, Pollen, Dust ,
,
Immune Support Supplement with Quercetin, Vitamin C, Zinc, Vitamin D3
GermGuardian Air Purifier with UV-C Light and HEPA 13 Filter, R emoves 99.97% of Pollutants
emoves 99.97% of Pollutants
HEPA Air Purifier, HEPA Air Cleaner with Washable Pre-Filter, AQI Display, ECO Mode, Sleep Mode, Pet Mode for Pets, Dust, Pollen, Removes 99.97% of Particles, Smoke, Mold, Pet Dander, Dust, Odors
Interesting Reads:
THE PREPPER'S CANNING & PRESERVING BIBLE: [13 in 1] Your Path to Food Self-Sufficiency. Canning, Dehydrating, Fermenting, Pickling & More, Plus The Food Preservation Calendar for a Sustainable Pantry
The Backyard Homestead: Produce all the food you need on just a quarter acre! P aperback
aperback
The Citizens' Guide to Geologic Hazards: A Guide to Understanding Geologic Hazards Including Asbestos, Radon, Swelling Soils, Earthquakes, Volcanoes
The Uninhabitable Earth: Life After Warming
Book: The Sixth Extinction:
An Unnatural History Paperback