There are affiliate links on this page. Read our disclosure policy to learn more.
 As greenhouse gas emissions from human activities increase,
they build up in the atmosphere and warm the climate, leading to many other
changes around the world-in the atmosphere, on land, and in the oceans. The
indicators in other chapters of this report illustrate many of these
changes. These changes have both positive and negative effects on people,
society, and the environment-including plants and animals. Because many of
the major greenhouse gases stay in the atmosphere for tens to hundreds of
years after being released, their warming effects on the climate persist
over a long time and can therefore affect both present and future
generations.
As greenhouse gas emissions from human activities increase,
they build up in the atmosphere and warm the climate, leading to many other
changes around the world-in the atmosphere, on land, and in the oceans. The
indicators in other chapters of this report illustrate many of these
changes. These changes have both positive and negative effects on people,
society, and the environment-including plants and animals. Because many of
the major greenhouse gases stay in the atmosphere for tens to hundreds of
years after being released, their warming effects on the climate persist
over a long time and can therefore affect both present and future
generations.
Many of the major greenhouse gases can remain in the atmosphere for tens to hundreds of years after being released. They become globally mixed in the lower atmosphere, reflecting contributions from emissions sources worldwide.
Several factors determine how strongly a particular greenhouse gas will affect the Earth's climate.
A second factor is each gas's unique ability to absorb energy.
By considering both of these factors, scientists calculate a gas's global warming potential, as compared to an equivalent mass of carbon dioxide (which is defined by a global warming potential equal to 1).
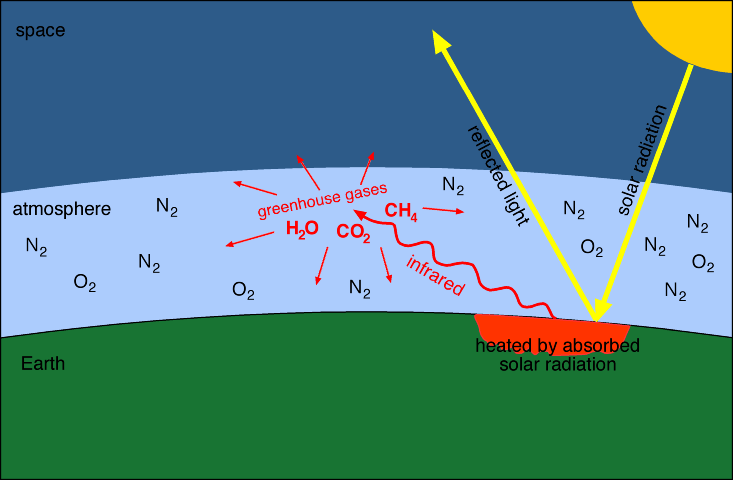
In a greenhouse, energy from the sun passes through the
glass as rays of light. This energy is absorbed by the plants, soil, and
other objects in the greenhouse. Much of this absorbed energy is converted
to heat, which warms the greenhouse. The glass helps keep the greenhouse
warm by trapping this heat.
The earth's atmosphere acts somewhat like
the glass of a greenhouse. About 31 % of the incoming radiation from the sun
is reflected directly back to space by the earth's atmosphere and surface
(particularly by snow and ice), and another 20 % is absorbed by the
atmosphere. The rest of the incoming radiation is absorbed by the earth's
oceans and land, where it is converted into heat, warming the surface of the
earth and the air above it. Particular gases in the atmosphere act like the
glass of a greenhouse, preventing the heat from escaping.
These
greenhouse gases absorb heat and radiate some of it back to the earth's
surface, causing surface temperatures to be higher than they would otherwise
be. The most important naturally occurring greenhouse gas is water vapour
and it is the largest contributor to the natural greenhouse effect. However,
other gases, although they occur in much smaller quantities, also play a
substantial and growing role in the greenhouse effect. These include carbon
dioxide, methane, and nitrous oxide.
Without this natural greenhouse
effect, the earth would be much colder than it is now - about 33 �C colder -
making the average temperature on the planet a freezing -18 �C rather than
the balmy 15 �C it is now. The warmth of our climate is crucial because on
earth and in the atmosphere, water can exist in all three of its phases -
frozen as snow or ice, liquid as water, and gaseous as water vapour. The
cycling of water from one phase to another is critical to sustaining life
since it is this cycling of water through the land-ocean-atmosphere system
that replenishes the water available to life on earth. The water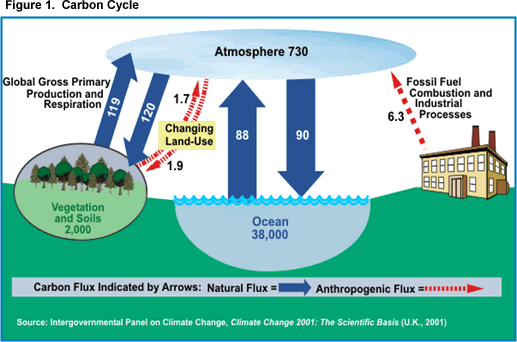 cycle is
also an important part of what drives our weather and the climate system
generally.
cycle is
also an important part of what drives our weather and the climate system
generally.
Iowa State University has an excellent summary: "Carbon dioxide and other greenhouse gases go through a natural cycle. The carbon cycle is shown in Figure 1. Large amounts of carbon pass back and forth between the atmosphere and the earth's surface. For example, growing crops and trees take in carbon dioxide (CO2) during photosynthesis. The carbon is the feedstock for making the plant and the oxygen (O2) is released into the atmosphere. When the plant dies and deteriorates or is processed, the carbon is combined with oxygen by microbial processes to become CO2 and is returned to the atmosphere. So these processes tend to keep the amount of carbon dioxide relatively constant over time."
This diagram provides a little more detailed explanation of sequestration of carbon dioxide emissions from a coal-fired plant:
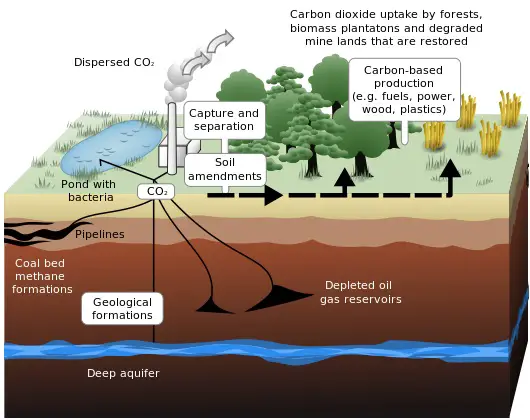
Schematic showing both terrestrial and geological sequestration of carbon
dioxide emissions from a coal-fired plant. Rendering by LeJean Hardin and
Jamie Payne. Source:
http://www.ornl.gov/info/ornlreview/v33_2_00/research.htm
Date 10 July
2009, 10:16 (UTC),
Government of Canada, Greenhouse effect diagram above
Columbia University, Greenhouse effect diagram diagram above
This page was updated on 25-Jan-2020